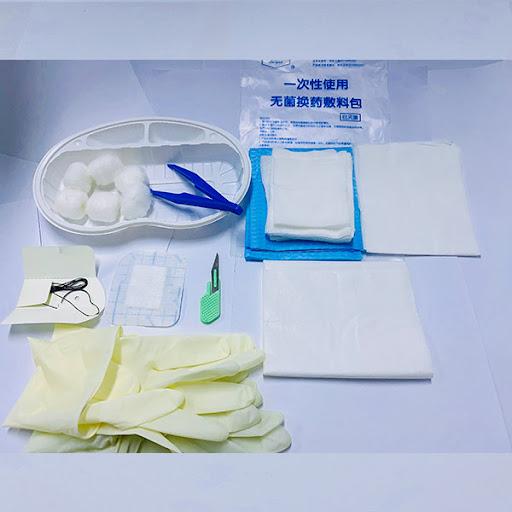
In today’s highly regulated medical supply industry, the role of a Dressing Kit Manufacturer has evolved far beyond simple assembly, becoming deeply integrated with sterilization science, precision packaging engineering, and global healthcare logistics systems that ensure safe delivery of wound care solutions across diverse clinical environments. This transformation reflects the growing demand for higher safety assurance and operational efficiency in both hospital and decentralized care settings.
Sterilization technology forms the foundation of modern dressing kit production. Medical-grade sterility is no longer achieved through a single method but through a carefully controlled multi-stage process that may include ethylene oxide treatment, gamma irradiation, or advanced plasma sterilization techniques depending on material compatibility. Each method is selected based on its ability to eliminate microbial contamination while preserving the physical integrity of sensitive dressing components. SUZHOU TEXNET CO., LTD. applies structured sterilization validation systems to ensure that every production batch maintains consistent microbial safety levels, supporting strict international healthcare requirements and reducing risks associated with cross-contamination in clinical use.
Packaging engineering plays an equally critical role in maintaining sterility throughout the product lifecycle. Modern dressing kits rely on multilayer barrier packaging systems designed to protect against moisture, oxygen, and external particulate intrusion. These packaging structures are engineered with precision sealing technologies that maintain integrity even under varying temperature and humidity conditions during transportation and storage. The internal layout of kits is also carefully optimized to ensure that healthcare professionals can access each component in a logical sequence without compromising sterile conditions. Within this engineering framework, the responsibilities of a Dressing Kit Manufacturer include not only product assembly but also the design of packaging systems that directly support clinical efficiency and infection control.
Automation has become a key driver in improving both production consistency and contamination control. Automated assembly lines reduce human contact with sterile components, significantly lowering the risk of environmental contamination during manufacturing. Robotics and sensor-based inspection systems are increasingly used to verify component placement, sealing accuracy, and packaging integrity in real time. This shift toward intelligent manufacturing also allows for higher scalability, enabling manufacturers to respond quickly to fluctuating global demand for medical consumables. SUZHOU TEXNET CO., LTD. integrates automation strategies into its production workflow to enhance precision while maintaining strict hygiene standards across all operational stages.
Regulatory compliance is another essential dimension shaping the development of dressing kit systems. Medical products must adhere to stringent international standards governing sterilization validation, material safety, and packaging performance. Compliance frameworks such as ISO quality systems and medical device regulations require manufacturers to implement comprehensive documentation, traceability, and quality assurance processes. This ensures that every kit can be traced back through its production lifecycle in the event of quality audits or safety reviews. In this environment, a Dressing Kit Manufacturer must maintain continuous alignment with evolving regulatory expectations while maintaining operational efficiency.
Logistics and supply chain management further influence how dressing kits are designed and distributed. Global healthcare networks depend on reliable delivery systems that preserve product sterility from manufacturing facilities to end users. Temperature stability, packaging durability, and transportation handling procedures all contribute to maintaining product integrity during distribution. Advanced tracking systems are increasingly being integrated into supply chains to monitor shipment conditions and ensure compliance with storage requirements. SUZHOU TEXNET CO., LTD. emphasizes coordinated supply chain strategies that support timely delivery while safeguarding product quality across international markets.
Innovation in material compatibility also supports the advancement of sterilization and packaging systems. As dressing kits incorporate more complex combinations of nonwoven fabrics, absorbent pads, and protective films, manufacturers must continuously adapt sterilization methods and packaging designs to accommodate diverse material behaviors. This requires close collaboration between material science teams and production engineers to ensure that all components maintain performance after sterilization processing. The integration of such interdisciplinary expertise allows manufacturers to develop more resilient and adaptable product systems.
Digitalization is also reshaping how production quality and supply chain performance are managed. Data-driven monitoring systems now track production variables such as sterilization exposure levels, packaging seal integrity, and environmental conditions in real time. These insights enable predictive quality control, reducing the likelihood of defects and improving overall operational transparency. As healthcare systems continue to demand higher reliability standards, the integration of digital monitoring becomes a critical advantage in maintaining global competitiveness.
The increasing complexity of modern healthcare environments continues to push dressing kit development toward greater precision, safety assurance, and system integration across manufacturing and logistics networks.https://www.sztexnet.com/product .